VSEPR Model
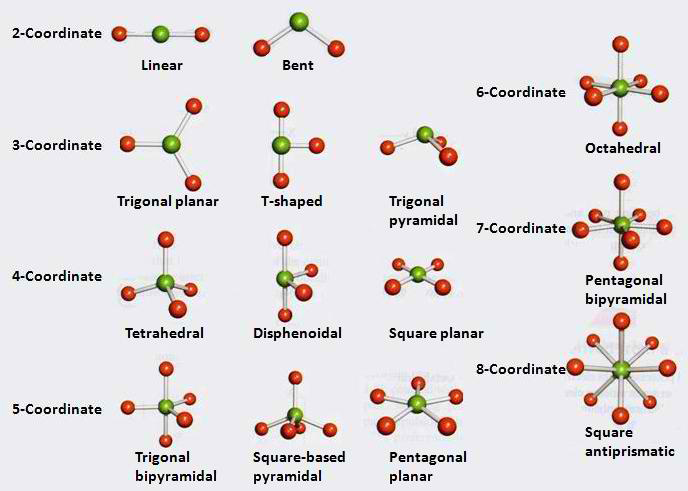
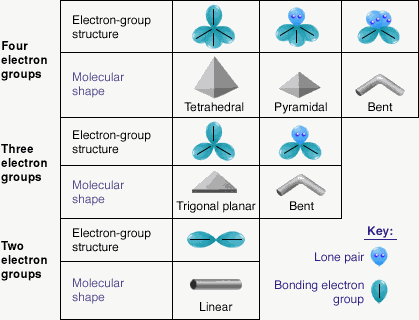
- The valence shell electron pair repulsion (VSEPR) model is useful for predicting the molecular structures of molecules formed from nonmetals
- The main idea of this model is that the structure around a given atom is determined by minimizing repulsions between electron pairs
- The bond and nonbonding electron pairs around a given atom are positioned as far apart as possible
- Whenever two pairs of electrons are present around an atom they should always be placed at an angle of 180 to each other to give
- Whenever three pairs of electrons are present around an atom, they should always be placed at the corners of a triangle in a plane at angles of 120 to each other
- Whenever four pairs of electrons are present around an atom, they should always be placed at the corners of a tetrahedron (the tetrahedral arrangement)