Because an atom has no overall charge, the number of electrons must equal the number of protons
James Chadwick discovered that the nuclei of most atoms contain neutrons as well as protons
All atoms of the same element contain the same number of protons and electrons, but atoms of a given element may have different numbers of neutrons
Isotopes - atoms with the same number of protons but different numbers of neutrons
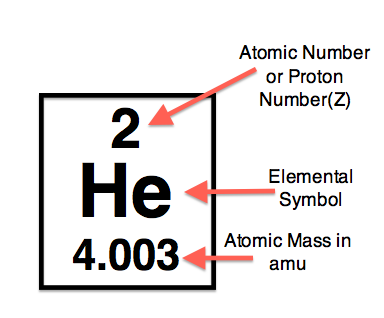
Atomic Number - the number of protons in a nucleus
Mass Number - the sum of the number of neutrons and the number of protons in a given nucleus
James Chadwick discovered that the nuclei of most atoms contain neutrons as well as protons
All atoms of the same element contain the same number of protons and electrons, but atoms of a given element may have different numbers of neutrons
Isotopes - atoms with the same number of protons but different numbers of neutrons
Atomic Number - the number of protons in a nucleus
Mass Number - the sum of the number of neutrons and the number of protons in a given nucleus