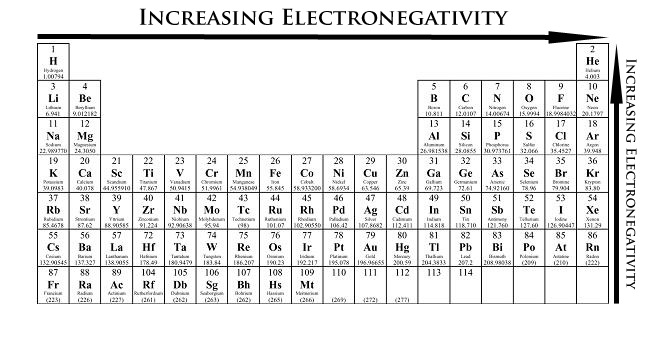
Electronegativity
- The unequal sharing of electrons between two atoms is described by electronegativity
- Electronegativity is the relative ability of an atom in a molecule to attract shared electrons to itself
- It increases going from left to right across a period and decreases going down a group for the representative elements
- The higher the atom's electronegativity value, the closer the shared electrons tend to be to that atom when it forms a bond