Chapter 14 - Change of Phases
Phase Changes: http://www.kentchemistry.com/links/Matter/PhaseChangesA.htm
Evaporation
Condensation
Fog and Clouds
Boiling
Freezing
Intramolecular forces - occur within the molecule
Intermolecular Forces - occur between molecules
Sublimation - going from a solid to a gas without going through a liquid stage
Deposition - going from a gas to a sold without going through a liquid stage
Crystalline Solids
Ionic Solids
Molecular Solids
Alloys
Heat of Fusion: http://hyperphysics.phy-astr.gsu.edu/hbase/thermo/phase2.html
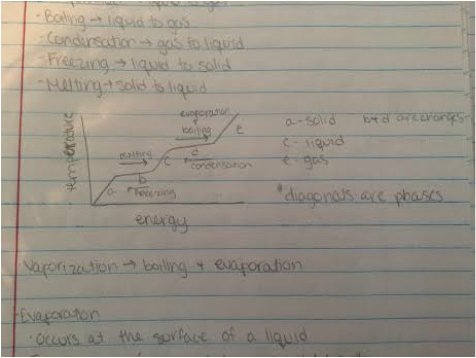
- Evaporation - liquid to gas
- Boiling - liquid to gas
- Condensation - gas to liquid
- Freezing - liquid to solid
- Melting - solid to liquid
- Vaporization - boiling and evaporation
Evaporation
- Occurs at the surface of liquids
- Temperature of any substance is related to the average kinetic energy of its particles
- Some particles have low kinetic energy and some have high kinetic energy
- When a molecule leaves, there is less energy in the system
- Cooling event
Condensation
- Kinetic energy ~ energy of attraction
- When gas molecules near the surface of a liquid are attracted to the liquid, they strik the surface with increased kinetic energy and become part of the liquid
- Heating event
Fog and Clouds
- Warm air rises and expands
- When the air expands it chills and the water molecules will slow down and stick together
- Formed when lots of molecules stick together
- When too much is attracted, it rains
Boiling
- Occurs everywhere in the liquid, not just the surface
- Boiling point - temperature at which a liquid boils; pressure is directly proportional; measured at standardized pressure
Freezing
- When kinetic energy is low and the energy of attraction is greater, a liquid will freeze
- Different liquids have different freezing points
Intramolecular forces - occur within the molecule
- Covalent - same
- Ionic - opposite
Intermolecular Forces - occur between molecules
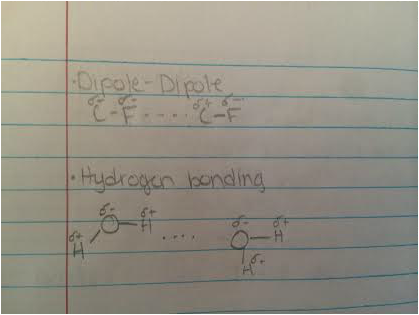
- Dipole-Dipole
- Hydrogen Bonding
- London Dispersion
Sublimation - going from a solid to a gas without going through a liquid stage
Deposition - going from a gas to a sold without going through a liquid stage
Crystalline Solids
- Ionic - ions as components
- Molecular - molecules as components
- Atomic - atoms as components
Ionic Solids
- High melting points
- Held together by a strong force existing between oppositely charged ions
Molecular Solids
- Low melting points
- Intermolecular forces are weak
- If molecules have a dipole moment, then the dipole-dipole forces hold solid together
- If molecules are nonpolar, then the London dispersion forces hold solid together
Alloys
- Substitutional
- Interstitial
- Mix between two metals
- http://www.youtube.com/watch?v=9LHDSB1n11k
Heat of Fusion: http://hyperphysics.phy-astr.gsu.edu/hbase/thermo/phase2.html