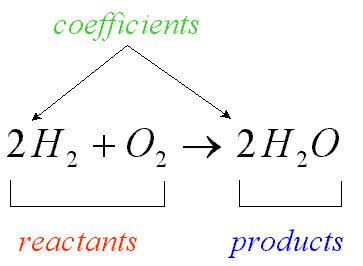Chemical Equations
- A chemical change always involves a rearrangement of the ways in which the atoms are grouped
- A chemical reaction is represented by writing a chemical equation in which the chemicals present before the reaction (reactants) are shown to the left of an arrow and the chemicals formed by the reaction (products) are shown to the right of an arrow
- The arrow indicates the direction of the change and is read as yields or produces
- The products contain the same atoms as the reactants but the atoms are associated in different ways
- In a chemical reaction, atoms are neither created nor destroyed
- There must be the same number of each type of atom on the product side as on the reactant side of the arrow
- The physical states of the reactants and the products are indicated in the equation by symbols
Definition of a spectator ion: http://www.definitionseek.com/chemistry/spectator_ion.html
(s) - solid
(l) - liquid
(g) - gas
(aq) - dissolved in water (in aqueous solution)