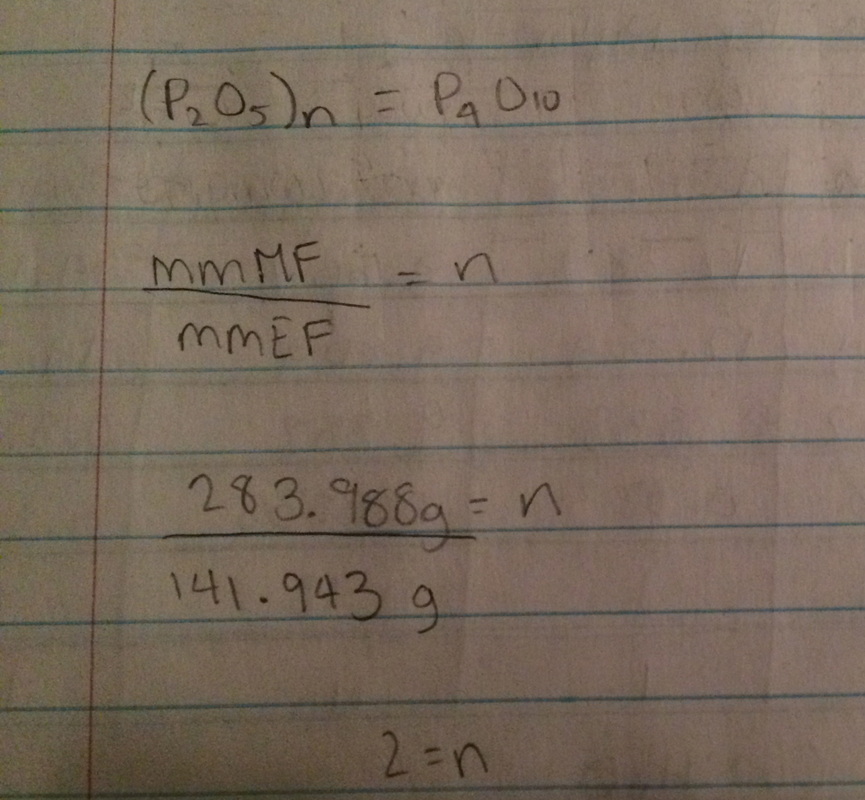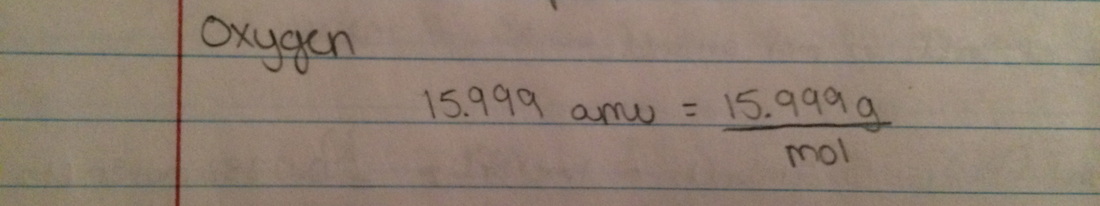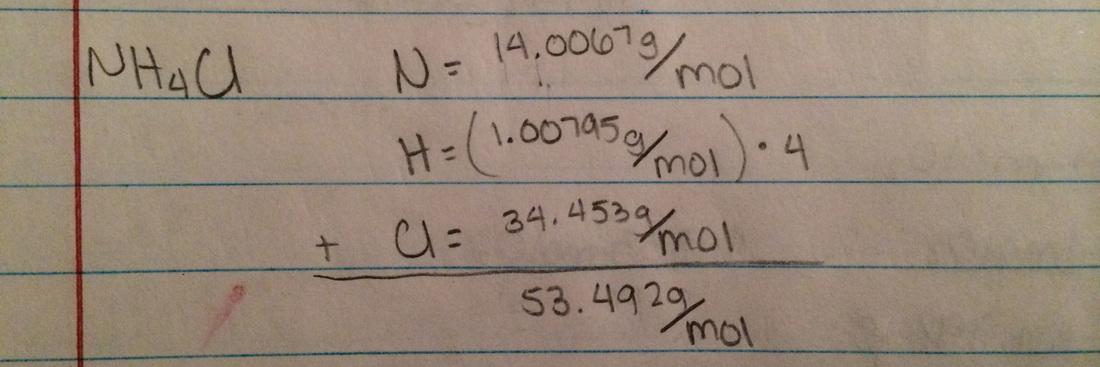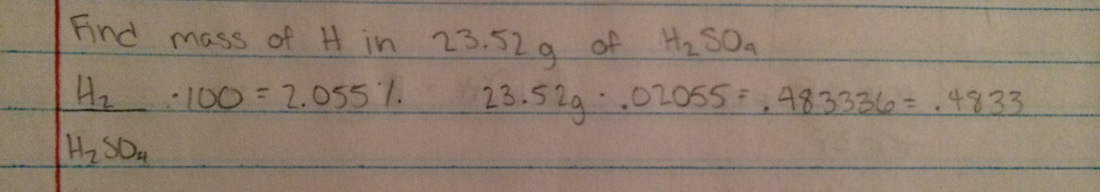Chapter 8 - Chemical Composition
Molar Mass - the mass of one mole in a substance
Finding the molar mass - http://www.occc.edu/kmbailey/chem1115tutorials/Stoichiometry_Molar_Mass.htm
Avrogadro's Number - 6.022*10^23 = 1 mol
Converting moles into atoms using Avrogadro's Number - http://antoine.frostburg.edu/chem/senese/101/moles/index.shtml
An empirical formula is already simplified and expresses the smallest whole-number ratio of the atom present
A molecular formula is not simplified and is the actual formula for a compound
More help on empirical and molecular formulas: http://www.ausetute.com.au/empirical.html
n has to be a whole number
Rounding Rules:
0.7 or greater --> round up
0.4 - 0.6 --> multiply by 2 (if you multiply one number by 2, you have to multiply all the numbers by 2)
0.0 - 0.3 --> round down
Activity on percent composition - http://www.yenka.com/activities/Percentage_Composition_by_Mass_-_Activity/
Finding the molar mass - http://www.occc.edu/kmbailey/chem1115tutorials/Stoichiometry_Molar_Mass.htm
Avrogadro's Number - 6.022*10^23 = 1 mol
Converting moles into atoms using Avrogadro's Number - http://antoine.frostburg.edu/chem/senese/101/moles/index.shtml
An empirical formula is already simplified and expresses the smallest whole-number ratio of the atom present
A molecular formula is not simplified and is the actual formula for a compound
More help on empirical and molecular formulas: http://www.ausetute.com.au/empirical.html
n has to be a whole number
Rounding Rules:
0.7 or greater --> round up
0.4 - 0.6 --> multiply by 2 (if you multiply one number by 2, you have to multiply all the numbers by 2)
0.0 - 0.3 --> round down
Activity on percent composition - http://www.yenka.com/activities/Percentage_Composition_by_Mass_-_Activity/