Chapter 13
Type 1 Nomenclature Naming Rules:
Practice Naming Type 1: http://quizlet.com/13509343/chemistry-quiz-1-nomenclature-flash-cards/
Type 2 Nomenclature Naming Rules:
Practice Naming Type 2: http://chemistry.about.com/od/testsquizzes/l/blionicnamesquiz.htm
Type 1 and 2 Naming rules: http://www2.pvc.maricopa.edu/tutor/chem/chem130/nomenclature/rules1.html
In type 1 compounds, the metal present forms only one type of cation
In type 2 compounds, the metal present can form two (or more) cations that have different charges
Type 3 Nomenclature Naming Rules:
Type 1 compounds are ionic compounds with metals that always form a cation with the same charge
Type 2 compounds are ionic compounds with metals (usually transition metals) that form cations with various charges
Type 3 compounds are compounds that contain only nonmetals
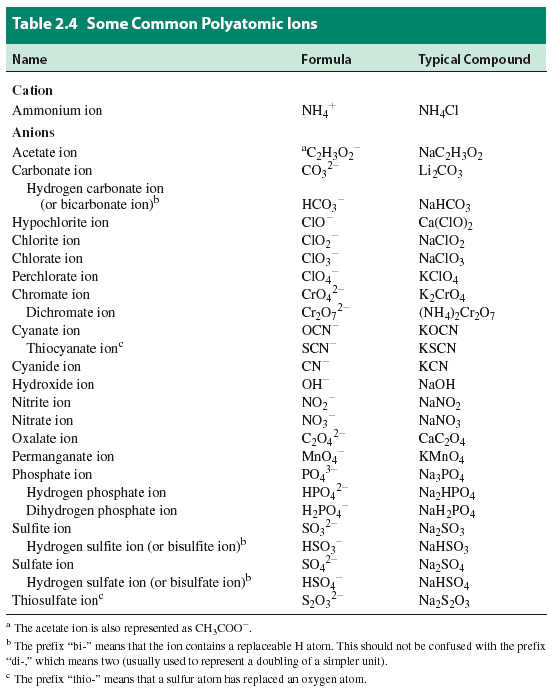
Polyatomic ions are charged entities composed of several atoms bound together
Video on Polyatomics: http://www.youtube.com/watch?v=p433lK8u5iw
When acids are dissolved in water, they produce H+ ions
An acid can be viewed as a molecule with one of more H+ ions attached to an anion
Acid Naming Rules:
Naming Acid Extra Rules: http://www.kentchemistry.com/links/naming/acids.htm
Naming Acids Video: http://www.youtube.com/watch?feature=player_embedded&v=27-aqIXf-Qg
- The Cation is always named first and the anion second
- A simple cation (obtained from a single atom) takes its name from the name of the element
- A simple anion (obtained from a single atom) is named by taking the first part of the element name (the root) and adding -ide
Practice Naming Type 1: http://quizlet.com/13509343/chemistry-quiz-1-nomenclature-flash-cards/
Type 2 Nomenclature Naming Rules:
- The cation is always named first and the anion second
- Because the cation can assume more than one charge, the charge is specified by a Roman numeral in parentheses
Practice Naming Type 2: http://chemistry.about.com/od/testsquizzes/l/blionicnamesquiz.htm
Type 1 and 2 Naming rules: http://www2.pvc.maricopa.edu/tutor/chem/chem130/nomenclature/rules1.html
In type 1 compounds, the metal present forms only one type of cation
In type 2 compounds, the metal present can form two (or more) cations that have different charges
Type 3 Nomenclature Naming Rules:
- The first element in the formula is named first, and the full element name is used
- The second element is named as though it were an anion
- Prefixes ar used to denote the numbers of atoms present
- The prefix mono- is never used for naming the first element
Type 1 compounds are ionic compounds with metals that always form a cation with the same charge
Type 2 compounds are ionic compounds with metals (usually transition metals) that form cations with various charges
Type 3 compounds are compounds that contain only nonmetals
Polyatomic ions are charged entities composed of several atoms bound together
Video on Polyatomics: http://www.youtube.com/watch?v=p433lK8u5iw
When acids are dissolved in water, they produce H+ ions
An acid can be viewed as a molecule with one of more H+ ions attached to an anion
Acid Naming Rules:
- If the anion does not contain oxygen, the acid named with the prefix hydro- and suffix -ic attached to the root name for the element
- When the anion contains oxygen, the acid name is formed from the root name of the central element of the anion or the anion name, with a suffix of -ic or -ous
Naming Acid Extra Rules: http://www.kentchemistry.com/links/naming/acids.htm
Naming Acids Video: http://www.youtube.com/watch?feature=player_embedded&v=27-aqIXf-Qg