Ionic Bonding and Structures of Ionic Compounds
- When metals and nonmetals react, the resulting ionic compounds are very stable
- The strong bonding in these ionic compounds results from the attractions among the oppositely charged cations and anions
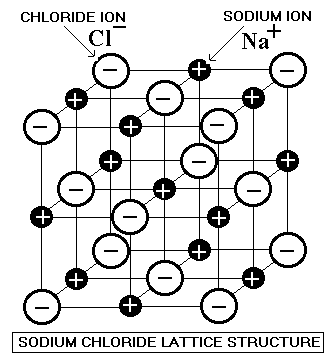
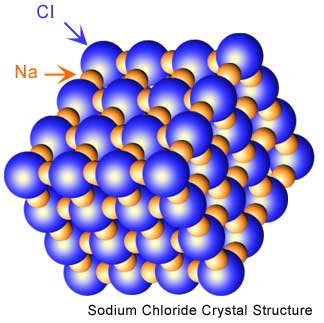
- The structures of virtually all binary ionic compounds can be explained by a model that involves packing the ions as though they were hard spheres
- The larger spheres (usually the anions) are packed together, and the small ions occupy the interstices (spaces or holes) among them
- To understand the packing of ions it helps to realize that a cation is always smaller than the parent atom, and an anion is always larger than the parent atom