Bond Polarity and Dipole Moment
- The most logical explanation for the development of bond polarity is hat the electrons in the bonds are not shared equally
- Bond Polarity - the partial positive and negative charges on the atoms in molecules
- We can account for the polarity of the HF molecule by assuming that the fluorine atom has a stronger attraction that the hydrogen atom for the shared electrons
- Bond polarity has important chemical implications
- Bond polarity is useful to assign a number that indicates an atom's ability to attract shared electrons
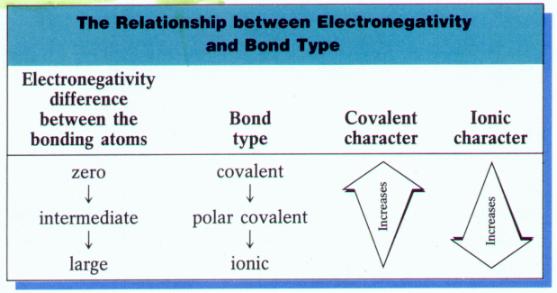
- The polarity of a bond depends on the difference between the electronegativity values of the atoms forming the bond
- If the atoms have very similar electronegativities, the electrons are shared almost equally and the bond shows little polarity
- The polarity of the bond increases as the difference in electronegativity increases
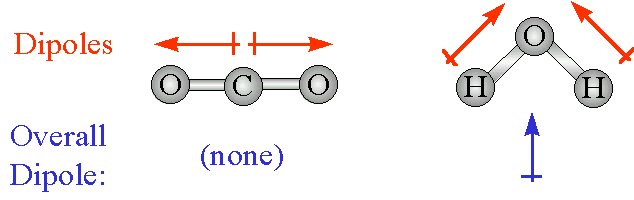
- A molecule that has a center of positive charge and a center of negative charge is said to have a dipole moment
- The dipolar character of a molecule is often represented by an arrow; this arrow points toward the negative charge center, and tis tail indicates the positive center of charge
- Any diatomic molecule that has a polar bond has a dipole moment; some polyatomic molecules also have dipole moments