Lewis Dot Structures
- Bonding involves just the valence electrons of atoms
- Valence electrons are transferred when a metal and a nonmetal react to form an ionic compound
- Valence electrons are shared between nonmetals in covalent bonds
- The Lewis Dot Structure is a representation of a molecule that shows how the valence electrons are arranged among the atoms in the molecule
- The rules for writing Lewis structures are based on observations of many molecules from which chemists have learned that the most important requirement for the formation of a stable compound is that the atoms achieve noble gas electron configurations
- In writing Lewis structures, we include only the valence electrons
- Hydrogen follows the duet rule
- When molecules obey the octet rule they are surrounded by eight electrons
- A single bond involves two atoms sharing one electron pair
- A double bond involves two atoms sharing two pairs of electrons
- A triple bond is one in which three electron pairs are shared
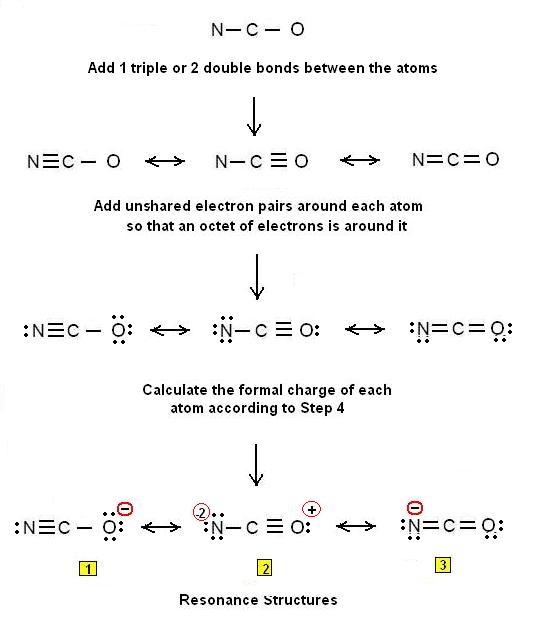
- A molecule shows resonance when more than one Lewis structure can be drawn for the molecule; in such cas we call the various Lewis structures resonance structures
Extra Practice: http://chemsite.lsrhs.net/bonding/flashLewis.html