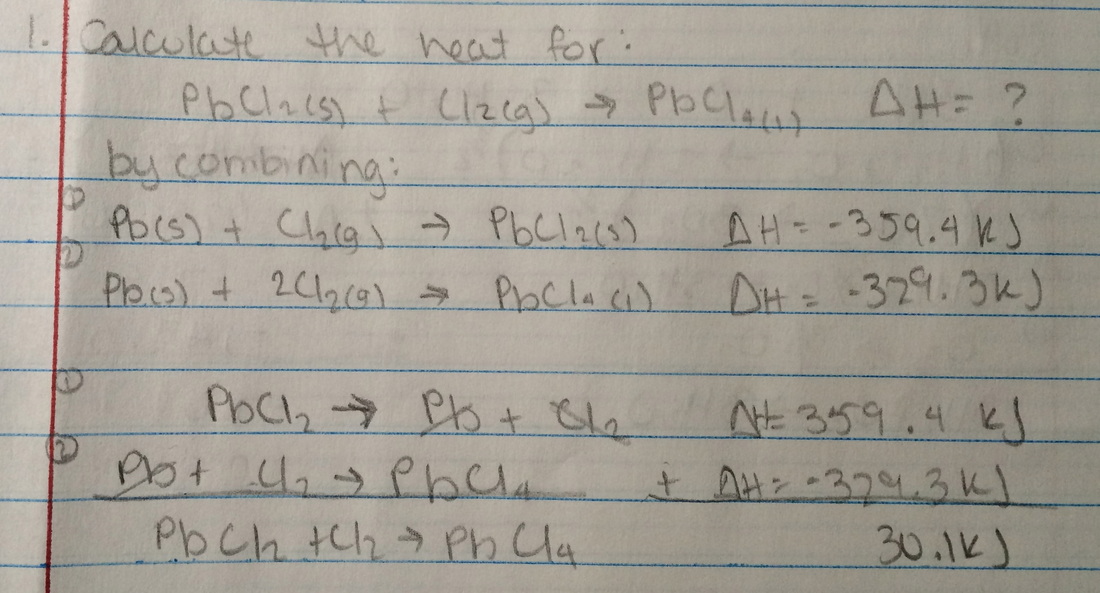Chapter 10 - Energy
Energy - the ability to do work
Potential Energy - stored energy
Kinetic Energy - the energy of motion
KE = (1/2)(m)(v^2)
It takes energy to release energy
Law of Conservation of Energy - energy can neither be created nor destroyed, only transferred
http://examples.yourdictionary.com/law-of-conservation-of-energy-examples.html
Force - the outcome of use of energy
Work = f*d
Temperature - measures energy (as heat)
Heat is the flow of energy
Heat flows from hot to cold naturally
Thermodynamic - study of energy
Internal energy: E=q+w
q=mc(Tfinal-Tinitial)
c=q/mT
Energy Units:
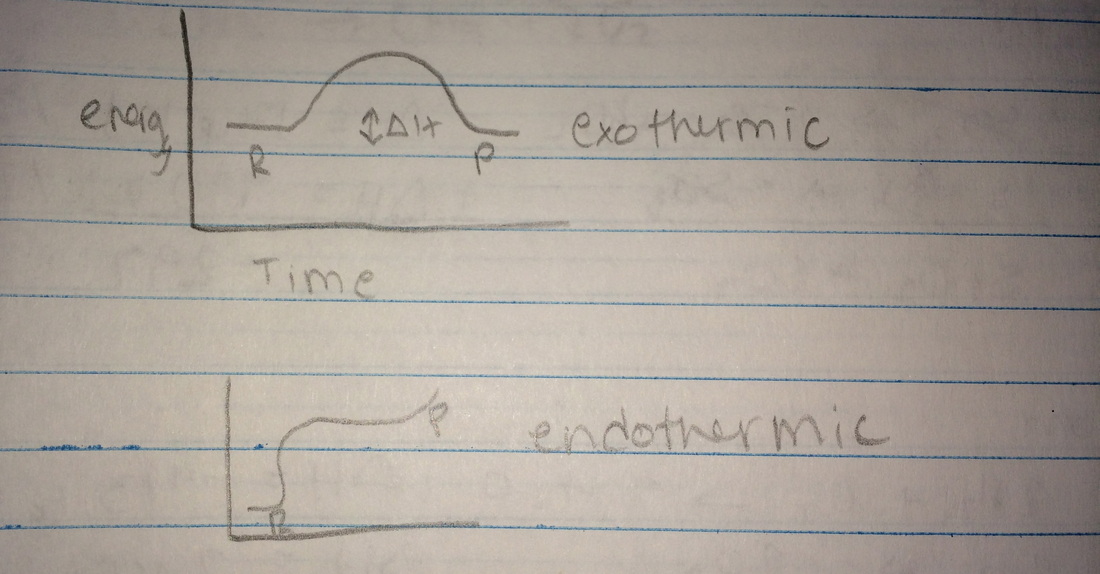
Enthalpy - flow of energy
Exothermic - energy is released from the system
Hess's Law (explained): http://www.science.uwaterloo.ca/~cchieh/cact/c120/hess.html
Hess's Law Activity: http://www.sciencegeek.net/Activities/Hesslaw.html
Potential Energy - stored energy
- Elastic - rubber bands
- Gravitational - goes against gravity
- Chemical - stored in any bonds
Kinetic Energy - the energy of motion
KE = (1/2)(m)(v^2)
It takes energy to release energy
Law of Conservation of Energy - energy can neither be created nor destroyed, only transferred
http://examples.yourdictionary.com/law-of-conservation-of-energy-examples.html
Force - the outcome of use of energy
Work = f*d
Temperature - measures energy (as heat)
- Kelvin - talks about energy (K=C+263.15)
- Celsius
- Fahrenheit (F=18*C+32)
Heat is the flow of energy
Heat flows from hot to cold naturally
Thermodynamic - study of energy
- First law: energy of the universe is constant because it flows between systems (this can be interchanged with the law of conservation of energy)
Internal energy: E=q+w
q=mc(Tfinal-Tinitial)
c=q/mT
Energy Units:
- Joules (J)
- Calories (Cal)
- 1 cal = 4.184 J
Enthalpy - flow of energy
- Exothermic - -Delta H
- Endothermic - +Delta H
Exothermic - energy is released from the system
Hess's Law (explained): http://www.science.uwaterloo.ca/~cchieh/cact/c120/hess.html
Hess's Law Activity: http://www.sciencegeek.net/Activities/Hesslaw.html