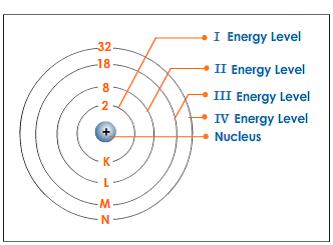
Energy Levels of Atoms
- An atom with excess energy is said to be in an excited state
- An excited atom can release some or all of its excess energy by emitting a photon and then moves to a lower energy state
- The lowest possible energy state of an atom is called its ground state
- Energy contained in the photon corresponds to the change in energy that the atom experiences in going from the excited state to the lower state
- Only certain energy levels can occur