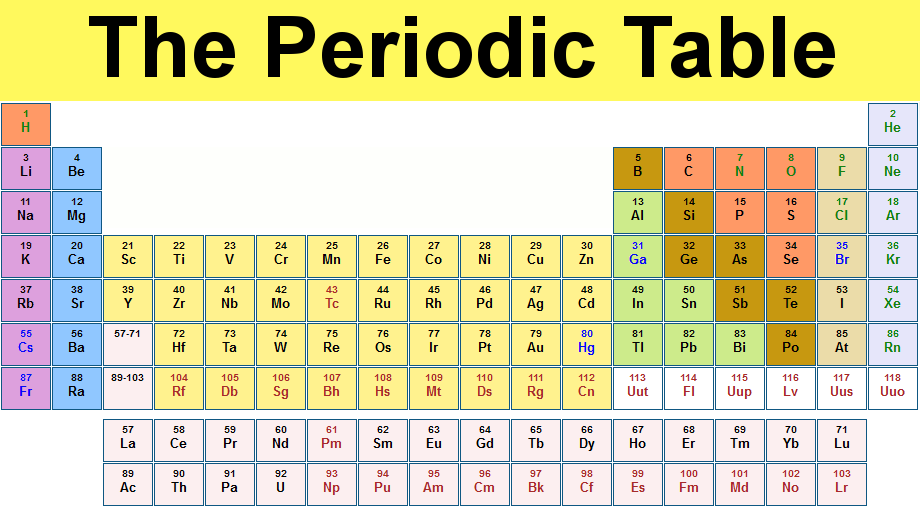
Periodic Table - a chart that chows all of the known elements
The number shown above each symbol is the atomic number for that element
Elements are listed in order of increasing atomic number
Groups - families of elements with similar chemical properties that lie in the same vertical column; often referred to by the number over the column
Alkali Metals - first column
Alkaline earth metals - group 2
Halogens - group 7
Nobel gases - group 8
Transition metals - large collection of elements that spans many vertical columns
Metals - most of the elements (efficient conduction of heat and electricity; malleability; ductility; lustrous appearance)
Nonmetals - upper right hand corner of the periodic table; generally lack those properties characterized with metals and show much more variation in their properties than metals do
Metalloids or Semimetals - silicon, germanium, arsenic, antimony, tellurium
The number shown above each symbol is the atomic number for that element
Elements are listed in order of increasing atomic number
Groups - families of elements with similar chemical properties that lie in the same vertical column; often referred to by the number over the column
Alkali Metals - first column
Alkaline earth metals - group 2
Halogens - group 7
Nobel gases - group 8
Transition metals - large collection of elements that spans many vertical columns
Metals - most of the elements (efficient conduction of heat and electricity; malleability; ductility; lustrous appearance)
Nonmetals - upper right hand corner of the periodic table; generally lack those properties characterized with metals and show much more variation in their properties than metals do
Metalloids or Semimetals - silicon, germanium, arsenic, antimony, tellurium